
About Cystinosis

About Cystinosis

About
Cystinosis
Cystinosis Overview
Cystinosis is a rare but serious multi-system genetic disorder that initially manifests in the kidneys during infancy and early childhood as renal Fanconi syndrome.1 A defect in the transport protein cystinosin causes free cystine to accumulate in the body, eventually forming crystals within bodily tissues.
Cystinosis is classified as a lysosomal storage disease and involves multiple organ systems.2 It is potentially sight-threatening.
Normal Lysosome

Cystinotic Lysosome

The most common form of cystinosis is caused by a deletion on the 57-kb segment of the CTNS gene, but over 100 molecular variants exist.2-4 As with other rare diseases, the complete diagnosis and detection of cystinosis is under-ascertained, leading to a delay in recognition.3 Severe (infantile) cases have severe mutations on both copies of the CTNS gene, while other patients may be heterozygous for the severe mutation. This variance is responsible for cystinosis presentations that are milder and/or late onset.
Clinically Recognized Phenotypes and Their Prevalence
Infantile (classic) nephropathic cystinosis
- Accounts for 95% of reported cases2,3
- Incidence at 1 per 100,000/200,000 live births worldwide2
- Approximately 600 affected children and adults in the U.S.5
Juvenile/late-onset (intermediate) nephropathic cystinosis
- Precise incidence unknown
- Same organ system involvement as nephropathic cystinosis, but with a slower progression of disease3
Non-nephropathic/ocular (benign) cystinosis
- Precise incidence unknown
- Characterized only by corneal crystal accumulation with no renal component or organ involvement whatsoever2



Nonrenal Complications of Cystinosis
- Retinal blindness
- Benign intracranial hypertension
- Central nervous system impairment
- Cerebral calcifications
- Corneal crystals
- Hypothyroidism
- Diabetes mellitus – requiring insulin therapy
- Pancreatic exocrine insufficiency
- Nodular regenerating hyperplasia of the liver
- Vacuolar myopathy
- Male hypogonadism
- Vascular calcifications
- Hypercholesterolemia
Nonrenal Complications
of Cystinosis
Ocular Complications of Cystinosis
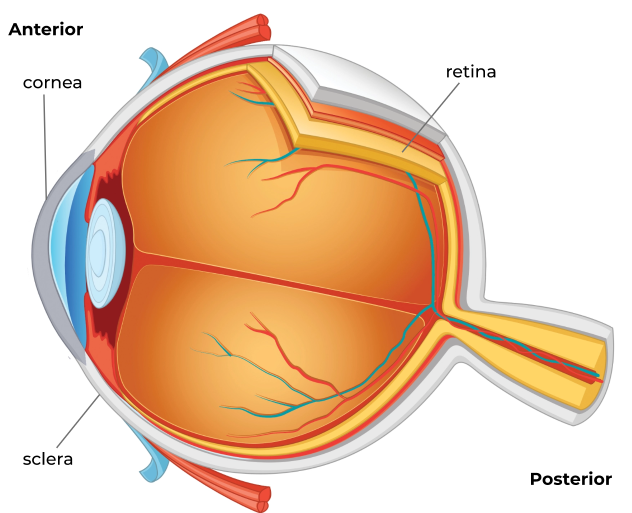
While a cystinosis patient’s systemic therapy can help prevent crystal accumulation in the eye’s posterior segment and prevent complications there, including retinal damage, there is no vascular supply to the cornea to deliver the drug.5,7,8

The cornea is left vulnerable to accumulating crystals, necessitating the use of a topical therapy.5,7,8 In untreated or undertreated cases, corneal complications can be severe8:


Diagnostic Methods and Criteria
Multiple methods exist for diagnosing cystinosis:

Systemic Diagnosis
Diagnosis of nephropathic cystinosis (whether infantile or juvenile) can be made by measuring leukocyte cystine content (LCC).3
- In unaffected persons, concentration is less than 0.2 nmol of half-cystine per mg of protein
- In nephropathic cystinosis patients, values exceed 2.0 nmol per mg of protein
Ocular Diagnosis
Imaging of the corneas may show crystal accumulation in affected patients and is a suitable diagnostic indicator for ocular cystinosis.5
- Slit-lamp photography is often used, although this is not a reliable method in infants younger than one year of age3
- Other ophthalmologic diagnostic tools are available
Molecular Diagnosis
Molecular diagnosis can be done in affected individuals to confirm the presence of the defective CTNS gene. This can be accomplished through prenatal chorionic villi sampling or DNA testing on cultured fibroblasts obtained from a skin biopsy.9
- Compound heterozygous or homozygous mutations should be found
- Genetic testing of both the patient and the patient’s family is recommended

Ophthalmic Treatment of Cystinosis

Clinical Management Considerations for Nephrologists and Other Non-Ophthalmic Specialists
If you have a cystinosis patient in your care, you should ask whether he or she is experiencing the following symptoms:

- Photophobia (typically the first and most commonly reported)7,10,11
- Blepharospasm (as a result of chronic squinting due to photophobia)10,11
- Chronic red eye7
- Foreign body sensation7
- Pain (which may be caused by recurrent corneal erosions or corneal scarring)7,8
Upon cystinosis diagnosis an ophthalmologist should be involved in patient care as soon as possible, and any treatment plan should encompass the entire body.7,8


Clinical Management Considerations for Ophthalmologists
In cystinosis patients with corneal crystal accumulation, prompt initiation of topical therapy is critically important.8 Left untreated, ocular complications are common causes of discomfort and disability in cystinosis patients.
- Even young children with corneal crystals can start to show signs of severe photophobia, including eye pain and difficulty opening the eyes in daylight12
- Topical therapy can reduce crystal density in patients of all ages, regardless of initial density10
- Corneal crystals accumulate if CYSTARAN is discontinued13

References: 1. Radojkovic, B. Cysteamine eye drops in the treatment of cystinosis – an Australian perspective. J Pharm Practice and Research 2015;45:440-445. 2. Nesterova G, Gahl WA. Nephropathic cystinosis: late complications of a multisystemic disease. Pediatr Nephrol 2008;23:863-878. 3. Gahl, W, Thoene JG, Schneider JA. Cystinosis. N Engl J Med. 2002;347(2):111-121. 4. Elmonem MA, Veys KR, Soliman NA, Van Duyk M, Van Den Heuvel LP, Levtchenko E. Cystinosis: a review. Orphanet J Rare Dis. 2016;11(47):1-17. 5. Huynh, N, Gahl WA, Bishop RJ. Cysteamine ophthalmic solution 0.44% for the treatment of corneal cystine crystals in cystinosis. Expert Rev. Ophthalmol. 2013;8(4): 341-345. 6. Gahl WA, Balog JZ, Kleta R. Nephropathic Cystinosis in Adults: Natural History and Effects of Oral Cysteamine Therapy. Ann Intern Med. 2007;147:242-250 7. Pinxten A-M, Hua M-T, Simpson J, Hohenfellner K, et al. Clinical Practice: A Proposed Standardized Ophthalmological Assessment for Patients with Cystinosis. Ophthalmol Ther. 2017;6:93-104 8. Bishop, R. Ocular Complications of Infantile Nephropathic Cystinosis. J Peds. 2017;183S:S19-S21. 9. Ariceta G, Camacho JA, Fernandez-Obsipo M, Fernandez-Polo A, et al. Cystinosis in adult and adolescent patients: Recommendations for the comprehensive care of cystinosis. Nefrologia. 2015;35(3):304-321. 10. Gahl WA, Kuehl EM, Iwata F, Lindblad A, Kaiser-Kupfer MI. Corneal Crystals in Nephropathic Cystinosis: Natural History and Treatment with Cysteamine Eyedrops. Molec Genet Metab. 2000;71:100-120. 11. Liang H, Baudouin C, Hassani RTJ, Brignole-Baudouin F, Labbe A. Photophobia and Corneal Crystal Density in Nephropathic Cystinosis: An In Vivo Confocal Microscopy and Anterior-Segment Optical Coherence Tomography Study. IOVS. 2015;56(5): 3218-3225 12. Tsilou E, Zhou M, Gahl WG, Sieving PC, Chan C-C. Ophthalmic Manifestations and Histopathology of Infantile Nephropathic Cystinosis: Report of a Case and Review of the Literature. Surv Ophthalmol. 2007;52(1):97-105. 13. CYSTARAN [prescribing information]. Gaithersburg, MD: Leadiant Biosciences, Inc.; Current Approved PI.
INDICATION & IMPORTANT SAFETY INFORMATION
CYSTARAN® (CYSTEAMINE OPHTHALMIC SOLUTION) 0.44% is a cystine-depleting agent indicated for the treatment of corneal cystine crystal accumulation in patients with cystinosis.
IMPORTANT SAFETY INFORMATION
- To minimize contaminating the dropper tip and solution, care should be taken not to touch the eyelids or surrounding areas with the dropper tip of the bottle. Keep bottle tightly closed when not in use.
- There have been reports of benign intracranial hypertension (or pseudotumor cerebri) associated with oral cysteamine treatment that has resolved with the addition of diuretic therapy. There have also been reports associated with ophthalmic use of cysteamine; however, all of these patients were on concurrent oral cysteamine.
- CYSTARAN contains benzalkonium chloride, which may be absorbed by soft contact lenses. Contact lenses should be removed prior to application of solution and may be reinserted 15 minutes following its administration.
- CYSTARAN is for topical ophthalmic use only.
- The most frequently reported ocular adverse reactions occurring in ≥ 10% of patients were sensitivity to light, redness, and eye pain/irritation, headache and visual field defects.
Please refer to CYSTARAN’s full Prescribing Information, available here.




